Navigating the Real World: A Scoping Review of Structured Frameworks to Effectively Identify, Evaluate, and Select Real-World Data Sources for Fit-for-Purpose Studies
Publisher: Clinical Pharmacology and Therapeutics Authors: Sonia Zebachi, Julien Tanniou, Elisabeth Bakker, Sieta T. de Vries, Rossella Di Bidino, Entela Xoxi, Anna Glaser, Gianluigi Savarese, Jan Hillert, Peter G.M. Mol, Kelly Plueschke, Billy Amzal, Ghinwa Y. Hayek, Jeverson Moreira View publication Abstract The potential of real-world data (RWD), particularly from patient registries, has been increasingly recognized […]
Navigating the Real World: A Scoping Review of Structured Frameworks to Effectively Identify, Evaluate, and Select Real-World Data Sources for Fit-for-Purpose Studies
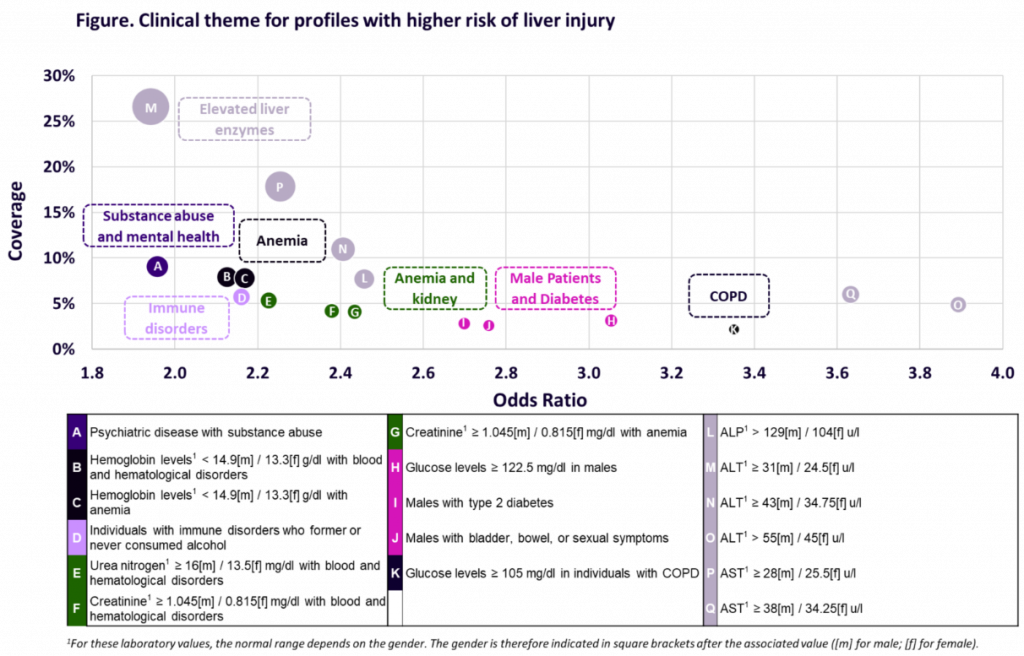
Event: EuroDURG 2025, Uppsala, Sweden Authors: Sonia Zebachi, Julien Tanniou, Elisabeth Bakker, Sieta T. de Vries, Rossella di Bidino, Entela Xoxi, Anna Glaser, Gianluigi Savarese, Jan Hillert, Peter G.M. Mol, Kelly Plueschke, Ghinwa Hayek, Billy Amzal, Jeverson Moreira View Poster The potential of real-world data (RWD), particularly from patient registries, has been increasingly recognised over […]
Decision-Making Criteria and Methods for Initiating Late-Stage Clinical Trials in Drug Development from a Multi-Stakeholder Perspective: A Scoping Review
Event: PSI 2025, Verona, Italy Authors: Ce Jiang, Céline Beji, Sonia Zebachi, Ghinwa Y. Hayek, Aysun Cetinyurek-Yavuz, Muhammad Bergas N. Fayyad, Laura Rodwell, Kit C. B. Roes, Billy Amzal, Christoph Gerlinger, Raphaël Porcher, Julien Tanniou View Abstract The decision-making process in drug development involves “go/no-go” decisions, particularly at the transition from early to late-stages trials. While the decisions are solely made by drug developers, they must […]
Decision-Making Criteria and Methods for Initiating Late-Stage Clinical Trials in Drug Development from a Multi-Stakeholder Perspective: A Scoping Review
Publisher: Clinical Pharmacology and Therapeutics Authors: Ce Jiang, Céline Beji, Sonia Zebachi, Ghinwa Y. Hayek, Aysun Cetinyurek-Yavuz, Muhammad Bergas N. Fayyad, Laura Rodwell, Kit C. B. Roes, Billy Amzal, Christoph Gerlinger, Raphaël Porcher, Julien Tanniou View Publication The decision-making process in drug development involves “go/no-go” decisions, particularly at the transition from early to late-stages trials. […]
On the Concepts, Methods, and Use of “Probability of Success” for Drug Development Decision-Making: A Scoping Review
Publisher: Clinical Pharmacology and Therapeutics Authors: Aysun Cetinyurek Yavuz, Muhammad Bergas Nur Fayyad, Ce Jiang, Florie Brion Bouvier, Celine Beji, Sonia Zebachi, Ghinwa Y. Hayek, Billy Amzal, Raphael Porcher, Julien Tanniou, Kit Roes, Laura Rodwell View publication Abstract Drug development is a lengthy process with considerable uncertainty at each milestone. Several trials are needed to progress […]

